Visualization
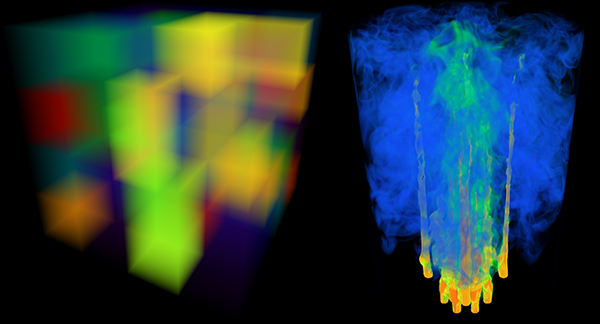
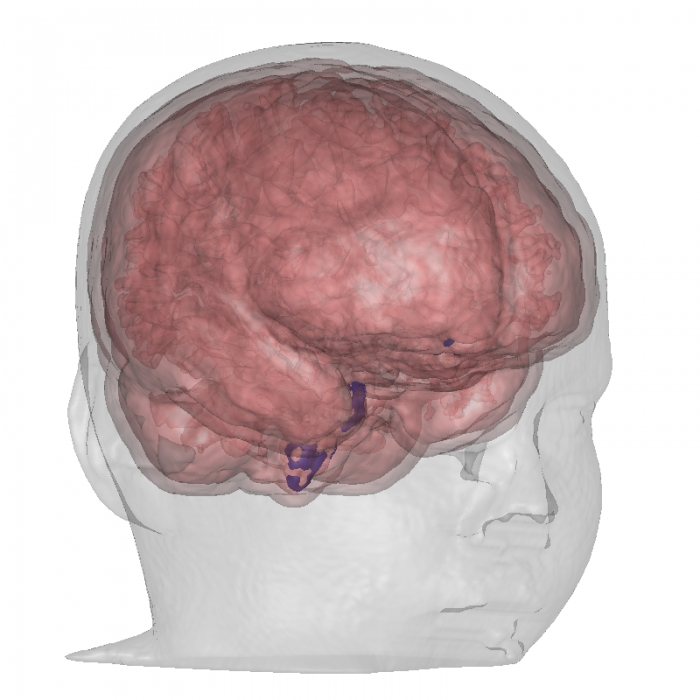
Visualization, sometimes referred to as visual data analysis, uses the graphical representation of data as a means of gaining understanding and insight into the data. Visualization research at SCI has focused on applications spanning computational fluid dynamics, medical imaging and analysis, biomedical data analysis, healthcare data analysis, weather data analysis, poetry, network and graph analysis, financial data analysis, etc.Research involves novel algorithm and technique development to building tools and systems that assist in the comprehension of massive amounts of (scientific) data. We also research the process of creating successful visualizations.
We strongly believe in the role of interactivity in visual data analysis. Therefore, much of our research is concerned with creating visualizations that are intuitive to interact with and also render at interactive rates.
Visualization at SCI includes the academic subfields of Scientific Visualization, Information Visualization and Visual Analytics.

Mike Kirby
Uncertainty Visualization
Alex Lex
Information VisualizationCenters and Labs:
- Visualization Design Lab (VDL)
- CEDMAV
- POWDER Display Wall
- Modeling, Display, and Understanding Uncertainty in Simulations for Policy Decision Making
- Topological Data Analysis for Large Network Visualization
Funded Research Projects:
Publications in Visualization:
  An Introduction to Verification of Visualization Techniques T. Etiene, R.M. Kirby, C. Silva. Morgan & Claypool Publishers, 2015. |
  Visualization C.R. Johnson. In Encyclopedia of Applied and Computational Mathematics, Edited by Björn Engquist, Springer, pp. 1537-1546. 2015. ISBN: 978-3-540-70528-4 DOI: 10.1007/978-3-540-70529-1_368 |
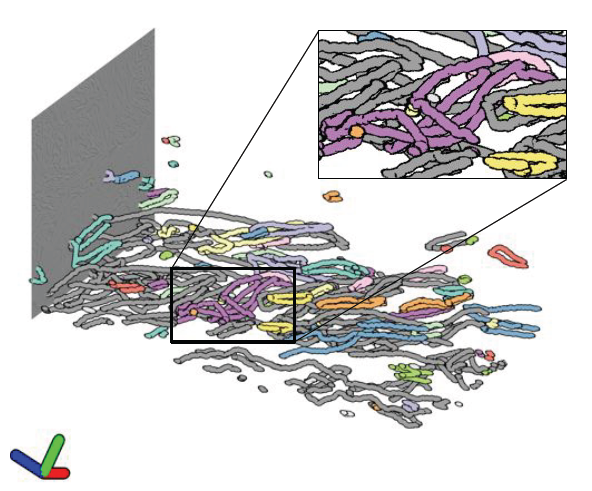
  CPU Ray Tracing Large Particle Data with Balanced P-k-d Trees I. Wald, A. Knoll, G. P. Johnson, W. Usher, V. Pascucci, M. E. Papka. In 2015 IEEE Scientific Visualization Conference, IEEE, Oct, 2015. DOI: 10.1109/scivis.2015.7429492 We present a novel approach to rendering large particle data sets from molecular dynamics, astrophysics and other sources. We employ a new data structure adapted from the original balanced k-d tree, which allows for representation of data with trivial or no overhead. In the OSPRay visualization framework, we have developed an efficient CPU algorithm for traversing, classifying and ray tracing these data. Our approach is able to render up to billions of particles on a typical workstation, purely on the CPU, without any approximations or level-of-detail techniques, and optionally with attribute-based color mapping, dynamic range query, and advanced lighting models such as ambient occlusion and path tracing. |
  Data Science: What Is It and How Is It Taught?, H. De Sterck, C.R. Johnson. In SIAM News, SIAM, July, 2015. |
 Visualization C.R. Johnson, K. Potter. In The Princeton Companion to Applied Mathematics, Edited by Nicholas J. Higham, Princeton University Press, pp. 843-846. September, 2015. ISBN: 9780691150390 |
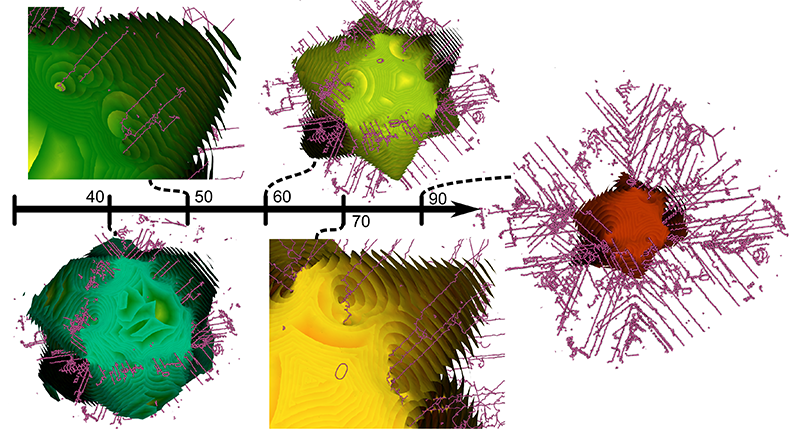
 Morse-Smale Analysis of Ion Diffusion for DFT Battery Materials Simulations, A. Gyulassy, A. Knoll, K. C. Lau, Bei Wang, P. T. Bremer, M. E. Papka, L. A. Curtiss, V. Pascucci. In Topology-Based Methods in Visualization (TopoInVis), 2015. Ab initio molecular dynamics (AIMD) simulations are increasingly useful in modeling, optimizing and synthesizing materials in energy sciences. In solving Schrodinger's equation, they generate the electronic structure of the simulated atoms as a scalar field. However, methods for analyzing these volume data are not yet common in molecular visualization. The Morse-Smale complex is a proven, versatile tool for topological analysis of scalar fields. In this paper, we apply the discrete Morse-Smale complex to analysis of first-principles battery materials simulations. We consider a carbon nanosphere structure used in battery materials research, and employ Morse-Smale decomposition to determine the possible lithium ion diffusion paths within that structure. Our approach is novel in that it uses the wavefunction itself as opposed distance fields, and that we analyze the 1-skeleton of the Morse-Smale complex to reconstruct our diffusion paths. Furthermore, it is the first application where specific motifs in the graph structure of the complete 1-skeleton define features, namely carbon rings with specific valence. We compare our analysis of DFT data with that of a distance field approximation, and discuss implications on larger classical molecular dynamics simulations. |
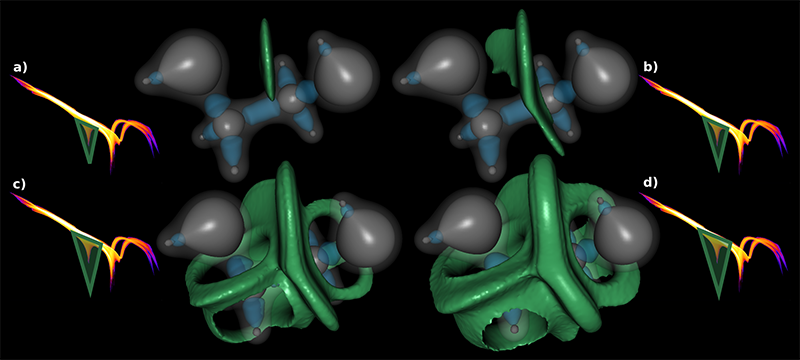
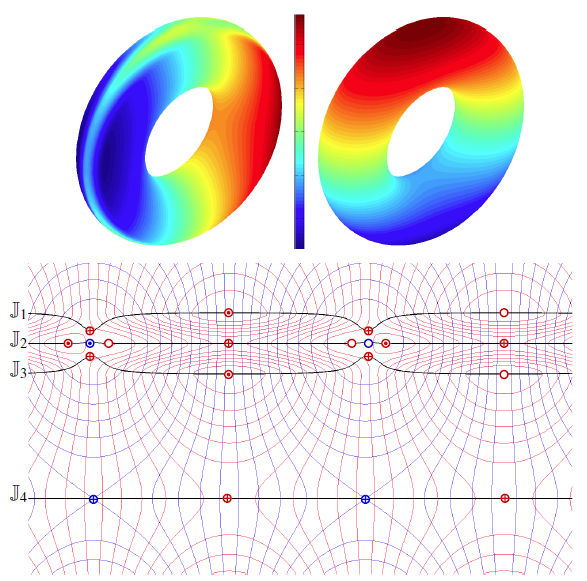
  Local, Smooth, and Consistent Jacobi Set Simplification H. Bhatia, Bei Wang, G. Norgard, V. Pascucci, P. T. Bremer. In Computational Geometry, Vol. 48, No. 4, Elsevier, pp. 311-332. May, 2015. DOI: 10.1016/j.comgeo.2014.10.009 The relation between two Morse functions defined on a smooth, compact, and orientable 2-manifold can be studied in terms of their Jacobi set. The Jacobi set contains points in the domain where the gradients of the two functions are aligned. Both the Jacobi set itself as well as the segmentation of the domain it induces, have shown to be useful in various applications. In practice, unfortunately, functions often contain noise and discretization artifacts, causing their Jacobi set to become unmanageably large and complex. Although there exist techniques to simplify Jacobi sets, they are unsuitable for most applications as they lack fine-grained control over the process, and heavily restrict the type of simplifications possible. |
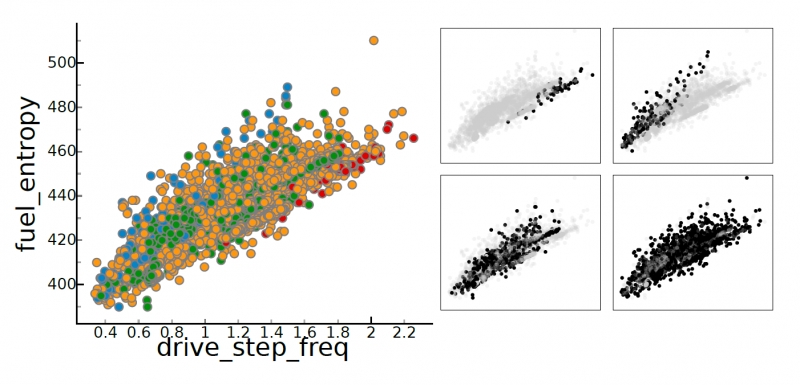
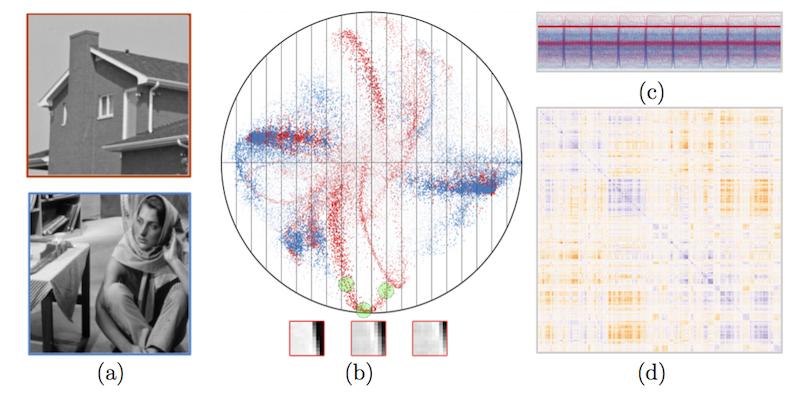
  Visual Exploration of High-Dimensional Data through Subspace Analysis and Dynamic Projections S. Liu, Bei Wang, J. J. Thiagarajan, P. T. Bremer, V. Pascucci. In Computer Graphics Forum, Vol. 34, No. 3, Wiley-Blackwell, pp. 271--280. June, 2015. DOI: 10.1111/cgf.12639 We introduce a novel interactive framework for visualizing and exploring high-dimensional datasets based on subspace analysis and dynamic projections. We assume the high-dimensional dataset can be represented by a mixture of low-dimensional linear subspaces with mixed dimensions, and provide a method to reliably estimate the intrinsic dimension and linear basis of each subspace extracted from the subspace clustering. Subsequently, we use these bases to define unique 2D linear projections as viewpoints from which to visualize the data. To understand the relationships among the different projections and to discover hidden patterns, we connect these projections through dynamic projections that create smooth animated transitions between pairs of projections. We introduce the view transition graph, which provides flexible navigation among these projections to facilitate an intuitive exploration. Finally, we provide detailed comparisons with related systems, and use real-world examples to demonstrate the novelty and usability of our proposed framework. |
  s-CorrPlot: An Interactive Scatterplot for Exploring Correlation, S. McKenna, M. Meyer, C. Gregg, S. Gerber. In Journal of Computational and Graphical Statistics, 2015. DOI: 10.1080/10618600.2015.1021926 The degree of correlation between variables is used in many data analysis applications as a key measure of interdependence. The most common techniques for exploratory analysis of pairwise correlation in multivariate datasets, like scatterplot matrices and clustered heatmaps, however, do not scale well to large datasets, either computationally or visually. We present a new visualization that is capable of encoding pairwise correlation between hundreds of thousands variables, called the s-CorrPlot. The s-CorrPlot encodes correlation spatially between variables as points on scatterplot using the geometric structure underlying Pearson's correlation. Furthermore, we extend the s-CorrPlot with interactive techniques that enable animation of the scatterplot to new projections of the correlation space, as illustrated in the companion video in Supplemental Materials. We provide the s-CorrPlot as an open-source R-package and validate its effectiveness through a variety of methods including a case study with a biology collaborator. |